Progress in Fe-N4O2 Single-Atom Catalysts for Antibiotic Wastewater Remediation
Editor: | Apr 26,2026
Antibiotic pollutants are highly persistent and refractory in aquatic environments, among which sulfamethoxazole (SMX) is a typical representative. Conventional water treatment technologies hardly achieve the deep mineralization of such contaminants. Heterogeneous Fenton-like reaction has drawn widespread attention, which adopts iron-based catalysts to activate hydrogen peroxide (H2O2) for generating strong oxidative reactive species. Nevertheless, traditional iron-nitrogen doped carbon (Fe-NC) catalysts commonly suffer from insufficient active site exposure, iron species aggregation, low H2O2 utilization efficiency and sluggish mass transfer, severely limiting their catalytic performance and practical application.
Research group from the Xinjiang Technical Institute of Physics and Chemistry (XTIPC), Chinese Academy of Sciences constructed a Fe-NC@MCM catalyst with distinctive sandwich hierarchical confinement structure to address the above bottlenecks. Further studies showed that the catalyst employed cetyltrimethylammonium bromide (CTAB) templated mesoporous silica (CTAB@MCM-41) as support. Via one-step pyrolysis strategy, it formed a multilevel composite structure containing inner hydrophilic carbon layer, middle mesoporous silica framework, and outer nitrogen-doped carbon shell embedded with atomically dispersed Fe-N4O2 coordination sites. X-ray absorption fine structure (EXAFS) analysis verified the isolated dispersion of Fe centers, which effectively suppressed metal aggregation and boosted active site accessibility as well as interfacial mass transfer. This work was published in Separation and Purification Technology.
In contrast to conventional iron-based catalysts with exposed unstable active sites, the hierarchically confined Fe-N4O2 motif endows the catalyst with ultra-fast activation capacity toward H2O2. In H2O2-based degradation system, nearly complete removal of SMX was realized within 4 min, with an apparent rate constant of 2.48 min-1. The total organic carbon (TOC) removal ratio reached 75%, accompanied by an ultrahigh H2O2 utilization efficiency up to 99%. Electron paramagnetic resonance (EPR) and radical quenching tests demonstrated that hydroxyl radicals (·OH) acted as the dominant reactive oxygen species. The entire degradation process consisted of hydroxylation, sulfonamide bond cleavage, aromatic ring nitration and ring-opening steps, finally mineralizing SMX into small inorganic products including CO2, H2O and SO42-. Moreover, the catalyst retained over 70% degradation efficiency after 5 consecutive cycles, with negligible iron leaching below the EU drinking water threshold (2 mg/L), exhibiting satisfying structural stability and environmental compatibility.
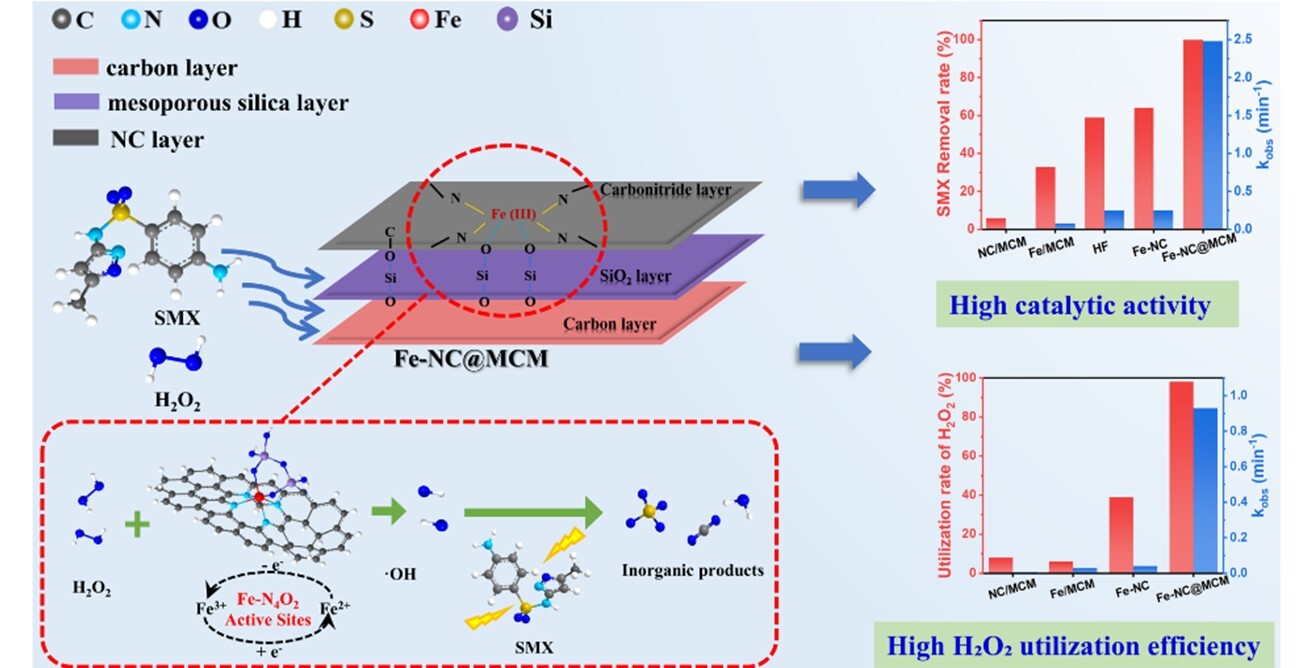
Figure 1 : Activation of H2O2over hierarchically confined sandwich-structured Fe-NC@MCM catalyst for SMX degradation with superior activity and ultrahigh H2O2 utilization efficiency. (image by XTIPC)
附件下载:
 (86) 991-3838931
(86) 991-3838931 lhskj@ms.xjb.ac.cn
lhskj@ms.xjb.ac.cn (86)991-3838957
(86)991-3838957 40-1 Beijing Road
Urumqi, XinjiangChina
40-1 Beijing Road
Urumqi, XinjiangChina