Progress in Iron-Based Biochar Catalytic Ozonation Combined with Micro-Nano Bubbles for Antibiotic Degradation
Editor: | Apr 26,2026
Sulfonamide antibiotics (e.g., sulfamethoxazole, SMX) are persistent and bioaccumulative in the aquatic environment, and their deep removal is difficult to achieve using conventional water treatment technologies. Heterogeneous catalytic ozonation (HCO) is a promising technique for degrading organic pollutants, as it can promote ozone decomposition to generate reactive oxygen species (ROS) via catalysts. However, HCO still faces two major bottlenecks in practical application: low ozone utilization efficiency, as well as insufficient catalytic activity and stability.Recently, the research group led by Prof. YIN J. at the Research Center for Energy and Chemical Engineering, Xinjiang Technical Institute of Physics & Chemistry, Chinese Academy of Sciences, addressed these challenges by designing and developing a nitrogen-doped cotton-straw-derived biochar catalyst supported with mixed-valent iron nanoparticles (FeOx-N-BC). By coupling this catalyst with ozone micro-nanobubbles (O3-MNBs), a dual-enhanced catalytic ozonation system (FeOx-N-BC/O3-MNBs) was constructed, enabling efficient degradation and mineralization of SMX. The relevant findings have been published in the Journal of Environmental Chemical Engineering, a prestigious journal in the field of environmental chemical engineering. This work provides a low-cost and highly stable strategy for the valorization of agricultural residues and the advanced treatment of antibiotic wastewater.
FeOx-N-BC This catalyst uses Xinjiang cotton straw as the carbon source and is prepared via impregnation and two-step pyrolysis. Nitrogen doping modulates the electronic structure, achieving uniform dispersion of iron active sites and coexistence of mixed-valent states Fe⁰/Fe²⁺/Fe³⁺, thus enhancing electron transfer and ozone activation efficiency. Meanwhile, ozone micro-nanobubbles strengthen gas–liquid mass transfer and significantly improve ozone dissolution and utilization efficiency.
Under optimal conditions, the FeOx-N-BC/O3-MNBs system achieves an SMX removal efficiency of 83.3%, a mineralization rate of 37.9%, and a pseudo-first-order rate constant of 0.30 min-1, outperforming single ozonation and undoped systems. EPR and quenching experiments verify that the system follows a radical–non-radical dual-pathway degradation mechanism dominated by singlet oxygen (1O2, contribution rate 25.13%) with hydroxyl radicals (·OH) as a secondary oxidant. XPS analysis confirms that the synergistic valence evolution of C, N, and Fe ensures sustained reactive oxygen species generation and catalytic stability.The catalyst retains 51.1% degradation efficiency after five cycles, exhibits strong resistance to common anions, and shows iron leaching below the detection limit, demonstrating outstanding stability and environmental safety.
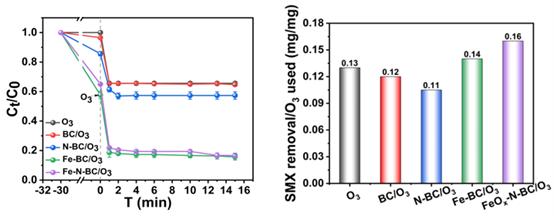
Figure 1 Removal efficiency of SMX and ozone utilization efficiency by catalytic ozonation with different catalysts
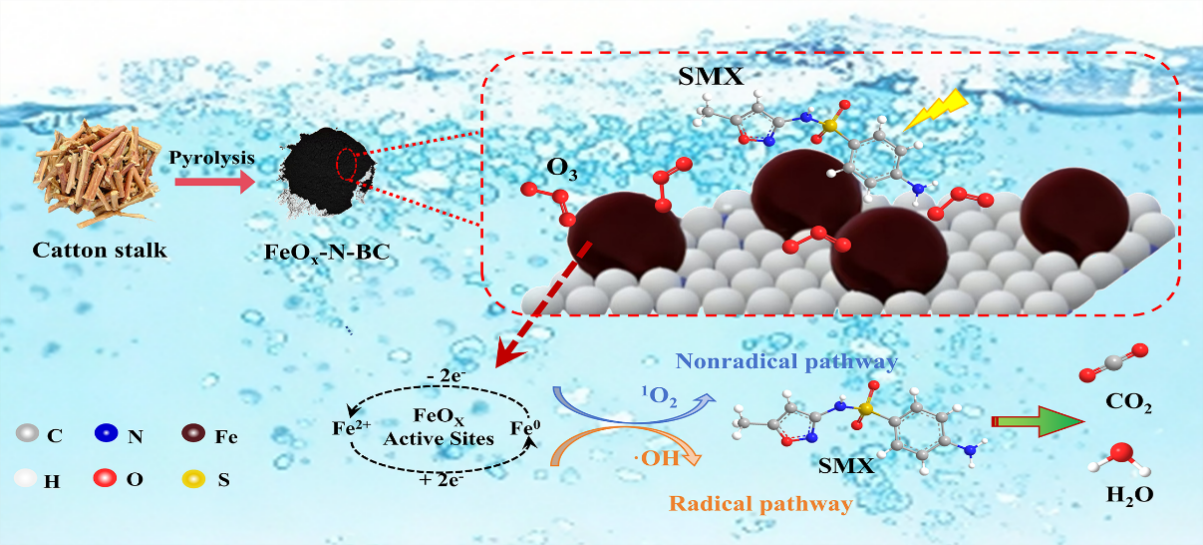
Figure 2 The specific reaction pathway and mechanism for the enhanced degradation of SMX by O3MNBs in the presence of FeOx-N-BC
附件下载:
 (86) 991-3838931
(86) 991-3838931 lhskj@ms.xjb.ac.cn
lhskj@ms.xjb.ac.cn (86)991-3838957
(86)991-3838957 40-1 Beijing Road
Urumqi, XinjiangChina
40-1 Beijing Road
Urumqi, XinjiangChina