Polycyclic aromatic hydrocarbons (PAHs), mainly produced via fossil fuel deposit and incomplete combustion, have been considered as one class of primary contaminants in naturally hydrophobic phases such as soil and sediment. Those organic molecules may undergo photolysis reactions, and this process is considered an important process for their fate in soil environment.
A research group led by Prof. WANG Chuanyi from Xinjiang Technical Institute of Physics & Chemistry (XTIPC), Chinese Academy of Sciences found the cation type influences both the direct and indirect photodegradation of pollutants on clay surfaces. Researchers explored photodegradation of PAHs (such as phenanthrene and anthracene) on smectite clays saturated with exchangeable cations possessing different propensities (i.e., Fe3+, Al3+, Ca2+, K+ and Na+) under various relative humidity (RH). Based on these results, researchers probed the role of interactions between PAHs and cation-smectite in PAHs transformation, and then evaluated the influence of surface properties of clay minerals on PAHs photodegradation.
Prof. WANG’s group revealed that the transformation of organic pollutants highly correlates to their adsorption activities on clay surfaces and highly dependent on the surfacial chemical and physical properties of the sorbent. Clay surfaces filled with different cations exhibited various physical-chemical properties and formation of different cation-π complexes.
Researchers showed complexation with stronger interaction characterizing by easier charge transfer, which induce higher direct photolysis rate of organic pollutants. It also can be found that the presence of various cations on clay surfaces influences the type and amount of reactive intermediates formation, which initiate the photodegradation of pollutants though indirect photolysis.
The study, published April 28 in the Journal of Hazardous Materials, gains insight into the mechanism responsible for PAHs photodegradation on clay surfaces and provides the basis for understanding the transformation and fate of PAHs in the natural soil environment. It also sheds light on the development of technologies for contaminated land remediation.
The work was supported by the National Natural Science Foundation of China, the Western Light Program of CAS, and the Hundred Talent Program of CAS etc.
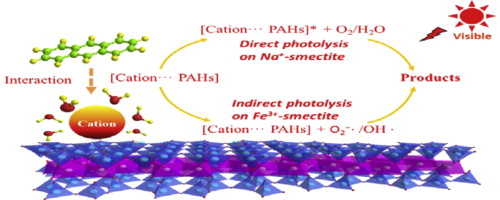
Roles of exchangeable cations in PAHs photodegradation on clay surafces under visible light(Image by XTIPC)
Contact:
Prof. WANG Chuanyi
E-mail: cywang@ms.xjb.ac.cn
Xinjiang Technical Institute of Physics & Chemistry
