In recent years, the emission of NOx is growing and the emitted NOx may lead to photochemical smog, acid rain, haze, etc. NOx mainly exists in the form of NO under high temperature conditions which accounts for about 95% of the initial released NOx. Generally, chemical removal of NO is mainly achieved through reduction or oxidation by catalysis. Oxidation is less desirable as it produces NO2 and NO3−.
Conventional approaches such as selective catalytic reduction, three-way catalysis, and wet scrubbing are not economically feasible to treat air pollutants in urban environments. Therefore, it is critical to develop a strategy to remove NO by selective reduction at very low concentrations.
A research group led by Prof. WANG Chuanyi at Xinjiang Technical Institute of Physics & Chemistry(XTIPC)of the Chinese Academy of Sciences developed a plasmonic Ag-TiO2 − x nanocomposite for the photocatalytic removal of NO with high selectivity under visible light. The study was published in Applied Catalysis B: Environmental.
Researchers used the commercial P25 (a mixture of anatase and rutile TiO2) as the raw materials to synthesize the visible-light-driven plasmonic photocatalyst Ag-TiO2 − x composite. The Ag loading amount was optimized at 2.5% (in mole ratio) based on former evaluation of the loading amount effects.
Results of electron paramagnanetic resonance (EPR) and Raman spectra prove that the post-annealing treatment of TiO2 (under oxygen-depleted condition) leads to oxygen loss and the formation of oxygen vacancies (VO).
High resolution transmission electron microscopy (HRTEM) results indicate that close Schottky contact was formed at Ag/TiO2 – x interface after the post-annealing treatment, which favors the photogenerated electrons transfer and restrains the carriers recombination.
Besides, researchers found the photoactivity of Ag-TiO2 − x composite was approximately twice higher than that of the commercial P25, and Ag-TiO2 − x could significantly inhibit the production of NO2.
Based on gas chromatography and Fourier transform infrared spectroscopy (FTIR) measurement, researchers deduced that the photo-oxidation of NO and selective photo-reduction of NO to N2 occur simultaneously during the process of NO removal by Ag-TiO2 − x. The oxidation of NO was due to the synergic effect between superoxide radicals (•O2−) and photogenerated holes (h+); while the selective photo-reduction was resulted from introduced VO in TiO2 − x.
This work provides a new insight into the different effects of photogenerated reactive species on NO photo-oxidation and VO on NO photo-reduction, and can open a promising avenue to developing novel materials with effective light harvesting property and reactivity for photocatalytic removal of NO.
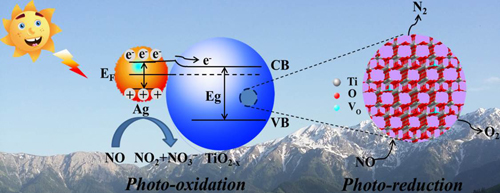
Figure: A plausible mechanism for visible light induced photocatalytic NO removal on Ag-TiO2 − x (Image by XTIPC)
Contact:
Prof. WANG Chuanyi
E-mail: cywang@ms.xjb.ac.cn
Xinjiang Technical Institute of Physics & Chemistry, CAS
