The artificial heterogeneous Z-scheme photocatalytic systems, mimicking the natural photosynthesis process, overcome the drawbacks of single-component photocatalysts with a wide light absorption range, long-term stability, high charge-separation efficiency and strong redox ability.
Hydrogen peroxide has been widely employed as energy reagent like rocket fuel or fuel in fuel cells. Previous reports have shown that some photocatalysts are capable of oxidizing H2O to O2 or OH- to •OH with the photogenerated holes, thus using H2O to replace organic scavenger to inhibit the recombination of photogenerated carries, meanwhile, preventing the H2O2 contamination. In addition, •OH may combine with each other to form additional H2O2, which open another channel of H2O2 formation.
A research group led by Prof. WANG Chuanyi at Xinjiang Technical Institute of Physics & Chemistry (XTIPC) of Chinese Academy of Sciences designed an all-solid-state Z-Scheme heterojunction photocatalyst (PIx-NCN) based on g-C3N4 nanosheets (NCN) to produce H2O2 via two-channel photocatalytic pathway, thus eliminating possible contamination from organic solvents and improving the production performance. The study was published in Journal of Catalysis
In fact, for the Z-Scheme photocatalyst (PIx-NCN), the photogenerated electrons and holes can be separated into two different phases, helping spatially isolate the oxidation and reduction reaction sites, and thus minimizing the catalytic deactivation.
Researchers found that more electrons from the conduction band of the NCN part for PIx-NCN are available to reduce O2 producing more H2O2, showing the improvement in H2O2 formation over that via the first channel. In addition, the valence band potential of PI is more positive than that of NCN, so the holes in valence band of PI for PIx-NCN can oxidize OH- to •OH and produce another part of H2O2 via the second channel.
Photocatalysis provides a potential strategy to solve environmental pollution problems by directly converting solar energy into electricity or chemical fuels, or decomposing organic pollutants under solar irradiation. This study provided a new strategy to improve the photocatalytic H2O2 production as well as to design the effective photocatalysts.
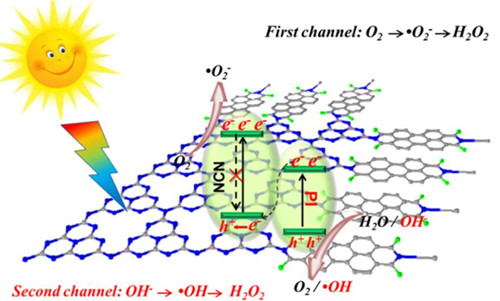
Figure: Schematic illustration of producing H2O2 via a two–channel pathway at different sites on all-solid-state Z-Scheme heterojunction. (Image by XTIPC)
Contact:
Prof. WANG Chuanyi
E-mail: cywang@ms.xjb.ac.cn
Xinjiang Technical Institute of Physics & Chemistry, CAS
