The Fenton reaction (Fe2+/H2O2) is one of most commonly used advanced oxidation processes, which can generate a highly reactive hydroxyl radical (•OH) for the degradation of organic pollutants. However, its application is limited by difficult separation and low recovery of the catalyst.
Recently, Fe3O4 was found to be an efficient and magnetic separation catalyst for the heterogeneous Fenton-like reaction. H2O2 is decomposed into •OH by the cycle involving the reduction state (Fe2+) and oxidation state (Fe3+). In the overall reaction, the reduction of Fe2+ to Fe3+ by oxidizing H2O2 is the rate-limiting step. Therefore, the rapid reduction of Fe3+ to Fe2+ is strongly desired for the Fenton-like catalytic process.
A research group led by Prof. ZHANG Yagang at Xinjiang Technical Institute of Physics & Chemistry (XTIPC) of Chinese Academy of Sciences designed a nanodispersed iron/graphene Fenton-like catalyst with high catalytic activity and reusability for catalytic removal of phenol. Researchers investigated the catalytic process and unveiled the mechanism of electron transfer facilitated regeneration of active Fe2+ by nanodispersed iron/graphene catalyst. The study was published in RSC advances.
Researcher used graphene oxide as precursor as support of catalyst, nano-scale Fe0 Nano particles (NPs) and Fe3O4 NPs were uniformly deposited on reduced graphene oxide (Fe0/Fe3O4-RGO). Fe0/Fe3O4-RGO achieved 100% removal efficiency for phenol within 30 min, and catalyst was reusable and the removal efficiency of phenol after 5 catalytic cycles was as high as 93%.
The Fe0 NPs and Fe3O4NPs were uniformly dispersed and distributed on RGO, furnishing more active sites. Fe2+ from the Fe3O4 and Fe0 of Fe0/Fe3O4-RGO reacted with H2O2 to generate •OH. Importantly, RGO served as an effective mediator to facilitate the electron transfer from Fe0 to Fe3+ for the regeneration of Fe2+, which speeded up the reduction of Fe3+ to Fe2+. In addition, the strong adsorption of phenol on the catalyst surface helped increasing the probability of the reaction between phenol and •OH formed at the surface of the catalyst.
The study provides new insight for the design of novel heterogeneous Fenton-like catalyst and the designed catalysts hold great potential application for removal of intractable organic pollutant.
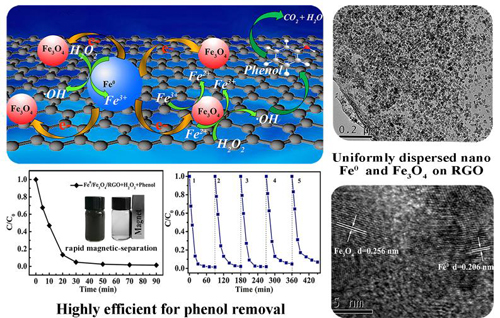
The mechanism of electron transfer facilitated regeneration of active Fe2+ by nano-dispersed iron/graphene catalyst for phenol removal(Image by XTIPC)
Contact:
Prof.ZHANG Yagang
E-mail: ygzhang@ms.xjb.ac.cn
Xinjiang Technical Institute of Physics & Chemistry, CAS
